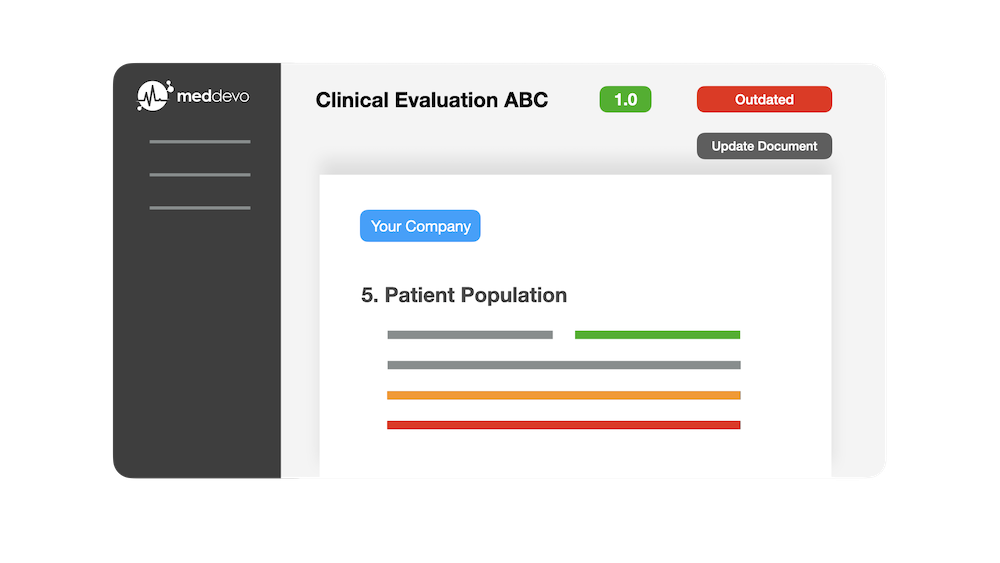
Automate your Technical Documentation
With meddevo eTD, teams get a workspace that allows each member to create, manage, and automate technical documentation for medical devices and IVDs. A turnkey SaaS.
40.000+ Medical Devices already managed in meddevo eTD








eTD Workflow

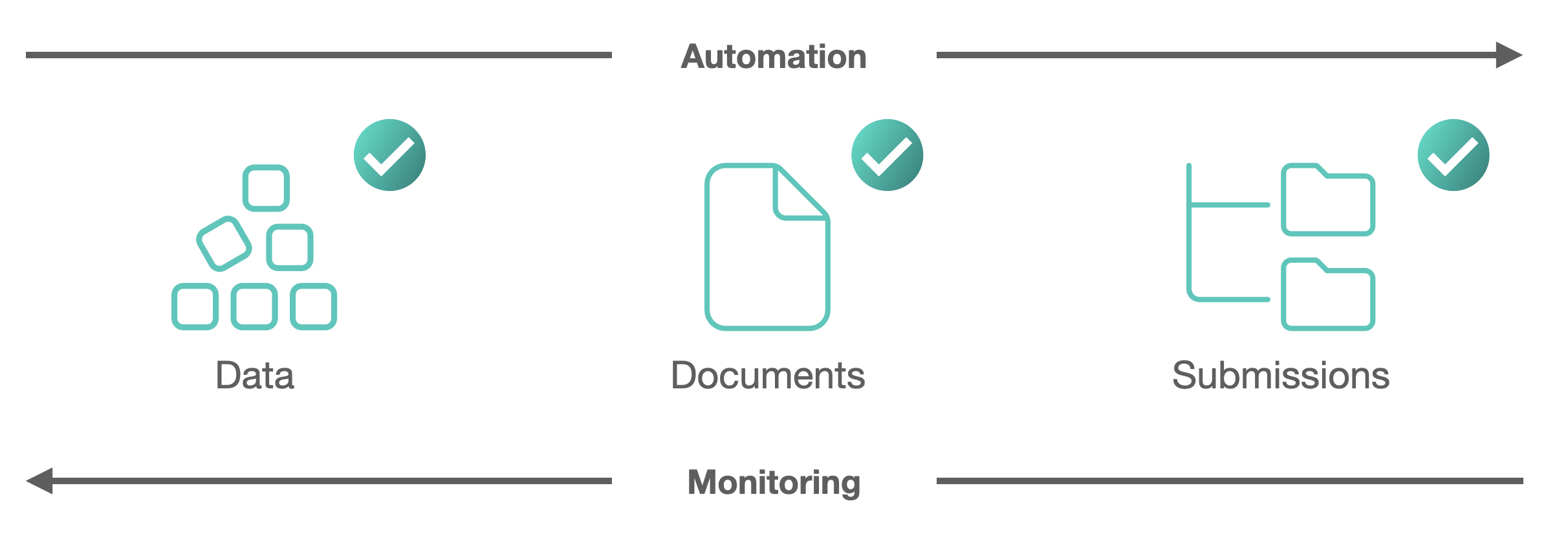

Sync your Quality with eTD
Your Technical Documentation on Autopilot
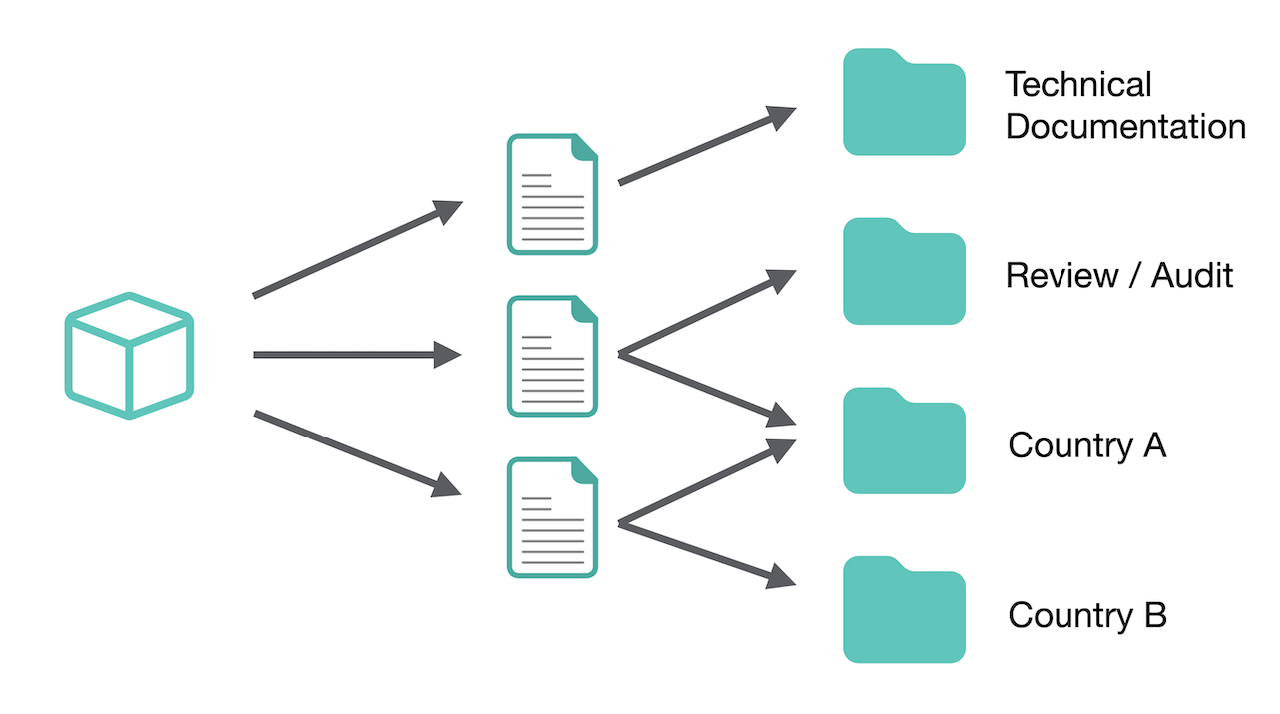


Lorem ipsum dolor sit amet, consetetur
Convallis volutpat elementum, scelerisque non. Nunc sed leo pellentesque eget. Ut commodo arcu suscipit proin at tellus. Cras habitant nec, venenatis, at hac feugiat consequat tristique pulvinar.

Convallis volutpat elementum, scelerisque non. Nunc sed leo pellentesque eget. Ut commodo arcu suscipit proin at tellus. Cras habitant nec, venenatis, at hac feugiat consequat tristique pulvinar.

Convallis volutpat elementum, scelerisque non. Nunc sed leo pellentesque eget. Ut commodo arcu suscipit proin at tellus. Cras habitant nec, venenatis, at hac feugiat consequat tristique pulvinar.

Convallis volutpat elementum, scelerisque non. Nunc sed leo pellentesque eget. Ut commodo arcu suscipit proin at tellus. Cras habitant nec, venenatis, at hac feugiat consequat tristique pulvinar.

In good company
Lorem ipsum dolor sit amet, consetetur
The fastest and easiest way to digitalize your
RA & QA










%20(1280%20%C3%97%20720%20px)-3.png)

